getting them excited
- Posted by darkeye on March 20th, 2008 filed in experiments, log
- Comment now »
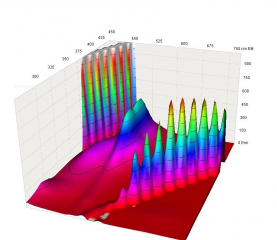
Today I made measurements with a phluorometer (or similarly called) machine, that can provide details on the excitation levels and the nature of fluorescence on my different bacteria. Fluorescence work in a way that one has to shine a specific light on the target, and alight of a different wavelength (color) will come out as a result.
It seems that for the TorA-GFPmut3* GFP, which we’re trying to control by pH change, the excitation wavelength (color) is just too close to the emitted light for a human eye to see.
The results basically show that the TorA-GFPmut3* GFP has a lower excitation point, which we’re seeing with our UV lights. But the one they are using in the Wilks article is around visible green (say around 500 nm), and the emitted light is around 525-545 nm. It seems that this is just too close to each other, or the level of fluorescent light coming out is just not high enough to see besides the original light that you put in.
Or I’m just colorblind 🙂
In the experiment, I’ve probably put too much of the TorA-GFPmut3* GFP bacteria into the test tube, thus the emitted light result overshoots a lot of time. But still, the excitation points are clearly visible.
I also measured the pGLO we have, which shows a healthy distance between excitation and emitted light: you put in light at around 375nm (non-visible), and you get light around 520nm (green) as a result. This strain has a lower excitation point as well, as the other one.




Leave a Comment
You must be logged in to post a comment.